アドホック調査
(患者・医師・医療従事者)
お客様のニーズに合わせて、医療用医薬品や医療機器の開発・マーケティングに活かせる
オリジナルの調査・分析をご提供します。
サービス概要
経験豊富なスタッフが、開発から上市後まで、プロダクトライフサイクルのあらゆる局面の意思決定について、マーケティングリサーチの視点からサポートします。
医療用医薬品や医療機器の市場は年々変化しています。コンセプト開発、承認申請、プロモーション立案など製品開発のあらゆる段階で市場ニーズを捉えた意思決定を行うことが成功の鍵となります。
こんな課題を解決できます
- 新製品の開発・導入を検討する際に市場性(市場規模)を評価したい
- 今後参入予定の市場で行われている治療や、治療に重要視されている要素を知りたい
- 患者視点でのPatient Journeyを把握し、アンメットニーズやレバレッジポイントを発見したい
- 開発中の新製品に対する医師の受容性を把握したい
- 既存製品に対する差別化ポイントを見つけ、新製品のポジショニングを明確にしたい
- 新製品のメッセージやビジュアルを決定したい
- 上市した製品が予定通り市場に浸透しているかを把握したい
- 上市後の売り上げが伸び悩んでいるため、原因を把握したい
- 適応を追加した場合の事業インパクトを定量的に知りたい
調査の流れ
調査の目的・アプローチしたい課題をお客様とすり合わせたうえで、最適な調査手法を選定し、企画から分析を一気通貫で行います。基本的な調査の流れは下記の通りです。
- 01企画立案
お客様のご事情やご要望を確認し、調査の目的・課題に応じた調査企画をご提案します。お客様による検討後、正式な調査依頼を受けてから、調査票などの作成を始めます。
- 02調査票の作成・準備
調査の目的・課題に見合った結果を導くための、調査票やディスカッションガイド(インタビューフロー)を作成します。調査票のドラフトをお客様と共に検討し、調査内容を確定します。
- 03調査実施・データ集計
合意した調査概要に従って、実際の調査(Webアンケートやインタビューなどのフィールドワーク等)を行います。
医師やコメディカルなどの医療従事者や患者などへのインタビュー調査は、医療分野専門の専属インタビュアーが実施します。フィールドワークの進捗状況の確認や、回収したデータのチェックを行い、質の高い情報の収集を行います。その後、回収した調査結果のデータを集計します。
- 04データの分析・報告書の作成
疾患領域や市場動向を熟知した担当者が、集計やデータ分析を実施します。報告書では、調査結果のサマリーに加え、調査目的に合わせたレコメンデーションをご報告します。
- 05プレゼンテーション
調査結果のプレゼンテーションを行います。調査結果に留まらず、当社で実施している自主企画調査のデータを踏まえて、ディスカッションすることも可能です。
実績
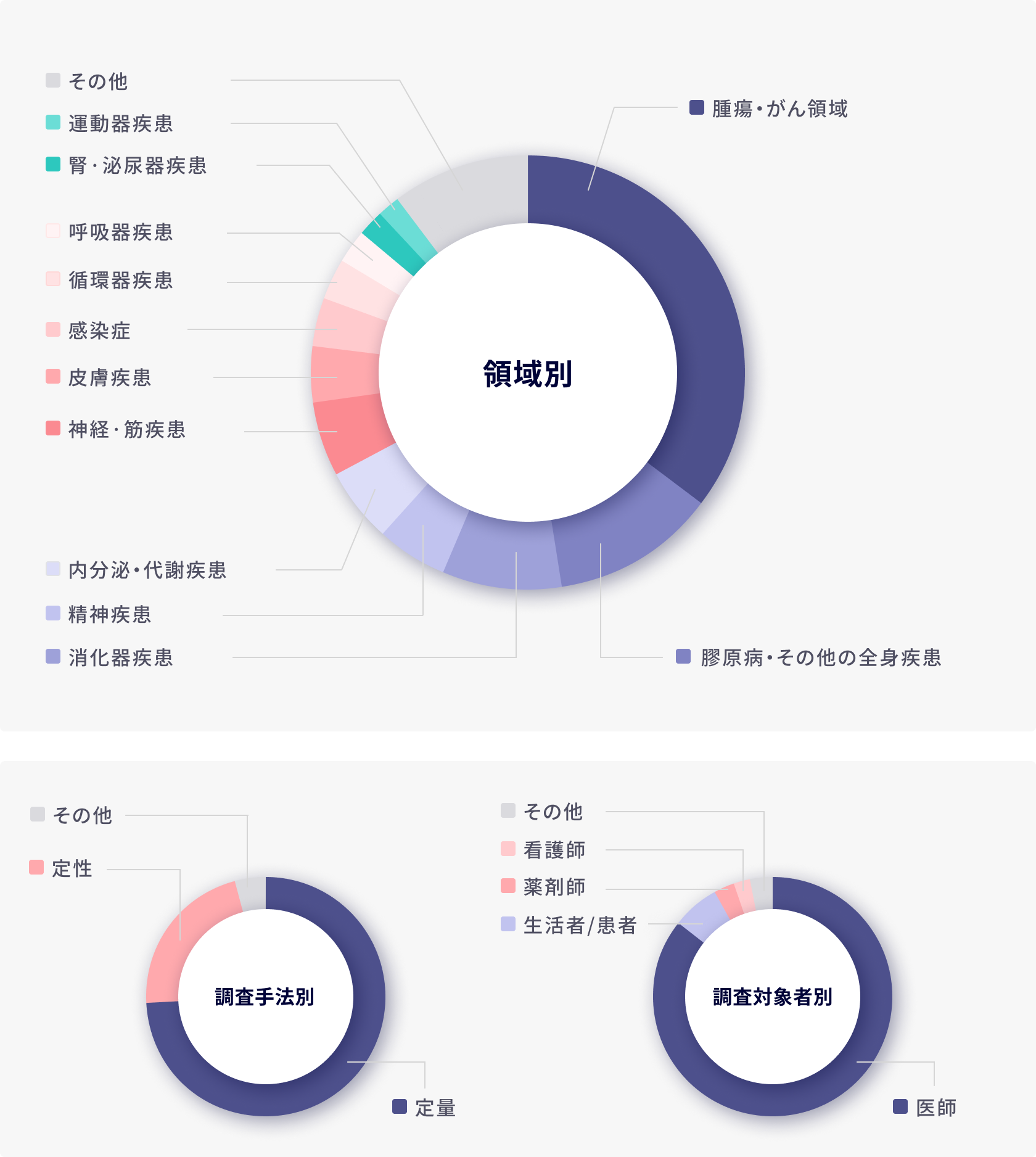
当社は年間約1,000本のプロジェクトを実施しており、医薬品関連では業界トップレベルのリサーチ実施経験を有しています。対象領域は糖尿病などのプライマリー領域、オンコロジー、希少疾患などのスペシャリティ領域に加え、治療用アプリを含む医療機器領域まで多岐に渡ります。
※ 「オンコロジー、希少疾患などのスペシャリティ領域」「治療用アプリを含む医療機器領域」は専門部署もございます。
また、医師、看護師、薬剤師、その他の医療従事者や患者及び介護者などのCaretakerも調査対象者に含むため、幅広いステークホルダーの調査を実施することが可能です。
2023年領域別受託実績(医療用医薬品・アプリ/医療機器)
インテージヘルスケアのエキスパート
長年の経験と、様々な疾患領域に専門性を持った担当者がプロジェクトをご支援します。
-

リサーチディレクター
加藤 明 KATO Akira
2001年から複数の調査会社において医療用医薬品のアドホック調査に従事。それ以前には、内資系製薬企業でMR経験あり。現場感も意識しながらマーケティング課題の解決に取り組んでいる。
-

リサーチディレクター
紫竹 宏亮 SHICHIKU Kosuke
2006年から医療用医薬品を中心に、アドホック・シンジケートや事業開発に従事。2023年にリサーチだけではない提案部署、2024年に医療機器専門部署を立ち上げ、部長として部門を率いる。
-

リサーチディレクター
土屋 晋 TSUCHIYA Shin
フィールド部門からキャリアをスタートし、これまで領域を問わず多くの市場調査を手掛けてきた。オンコロジー関連学会への参加や製薬企業へのオンサイト勤務を経験。クライアントが抱える様々な課題解決に応えている。
調査事例
具体的な調査の事例を紹介します。お客様の課題に合わせ、柔軟な調査手法のご提案・実施・分析を行っています。
- マーケティングリサーチ
新製品上市前の一連の調査を提案
- 新製品
- 新領域
企業・組織- 医療用医薬品
- マーケティングリサーチ
ターゲット世代に刺さる疾患啓発をデータでサポート
- 疾患啓発
- 患者視点
- ターゲティング
- ペルソナ
企業・組織- 医療用医薬品
- マーケティングリサーチ
医療機器プロモーショントラッキングで市場を把握
- 医療機器
企業・組織- 医療機器・アプリ
よくある質問
お客様へのヒアリングをベースに進めていくことも可能であるため、必ずしも事前にご準備いただく必要はありません。調査目的や現在の課題を最初にお伺いし、調査課題の整理から調査へ落とし込む方法を含めてご支援いたします。お困りごとがあれば、まずはお問い合わせください。
調査の目的によって異なります。当社では、医師や看護師、薬剤師などの医療従事者だけでなく、患者や介護者などの医療消費者を含め、広く医療に関わるステークホルダーを対象とした調査が可能です。お客様の調査目的に合わせて、適切な対象者やリクルート条件をご提案します。
当社の自主企画調査や過去経験から、適切な属性や選定条件をご提案いたします。また、獲得可能医師サンプルサイズもあわせてご提案いたします。詳しくはお問い合わせください。